
9/17/2020
PITTSBURGH – UPMC Hillman Cancer Center is the only cancer center in western Pennsylvania, and among a select few cancer centers in the nation, certified to offer the latest immunotherapy for patients with mantle cell lymphoma (MCL).
Recently approved by the U.S. Food and Drug Administration (FDA), TECARTUS™ (brexucabtagene autoleucel), is the first and only chimeric antigen receptor (CAR) T-cell therapy for the treatment of adults with relapsed or refractory MCL. This is a rare form of non-Hodgkin lymphoma that originates from cells in the “mantle zone,” or outer edge of the lymph node. It also is highly aggressive following relapse, with many patients progressing following therapy.
UPMC Hillman Cancer Center was first in the region to offer CAR T-cell therapies, a cancer treatment that uses cells from a patient’s own immune system. Since then, Hillman has expanded this treatment with both FDA-approved and investigational CAR-T therapies for other blood cancers, including leukemia, lymphoma and multiple myeloma.
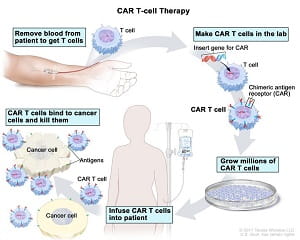 CAR T-cell therapy involves extracting a type of white blood cells—or T cells—from a patient and then genetically engineering them to become CAR T cells so they can recognize and destroy cancer cells when they are infused back into the patient.
CAR T-cell therapy involves extracting a type of white blood cells—or T cells—from a patient and then genetically engineering them to become CAR T cells so they can recognize and destroy cancer cells when they are infused back into the patient.
“This is an exciting new commercially available treatment option for our patients with mantle cell lymphoma, which takes a similar approach to what already has been so successful in treating relapsed and refractory diffuse large B cell lymphomas,” said Warren Shlomchik, M.D., director of the hematopoietic stem cell transplant and cell therapy programs at UPMC Hillman Cancer Center. “We also look forward to new approvals for other adoptive T cell immunotherapies in the near future.”
To date, more than 100 CAR T-cell therapies have been administered, which is seven times more than any other institution in western Pennsylvania.
“We have been leaders in the development of immunotherapy treatments for several decades at Hillman, and it certainly is rewarding for us to be among the top cancer centers in the country to continually offer new advancements for our patients as they become available,” said Robert Ferris, M.D., Ph.D., director of UPMC Hillman Cancer Center. “We have dedicated teams of doctors, nurses, pharmacists and staff who have been involved for several years and are well prepared.”
UPMC Hillman Cancer Center is the region’s only comprehensive cancer center designated by the National Cancer Institute, and one of just 52 such centers in the country. Hillman also has one of the largest cancer networks, with nearly 80 centers in four states and internationally, making access to new therapies and clinical trials available to patients close to home.
“Our oncologists, hematologists and researchers, who work side by side at Hillman, are dedicated to finding new treatment options for our patients when other treatments have failed them,” said Stanley Marks, M.D., chairman, UPMC Hillman Cancer Center. “What’s also important is that we can provide these new advances for cancer patients throughout the Hillman network.”
To learn more about CAR T-cell therapy and other cancer research advancements, visit the UPMC Hillman Cancer Center website.
PICTURE INFO: Click image for high-resolution version.
CREDIT: The National Cancer Institute
















