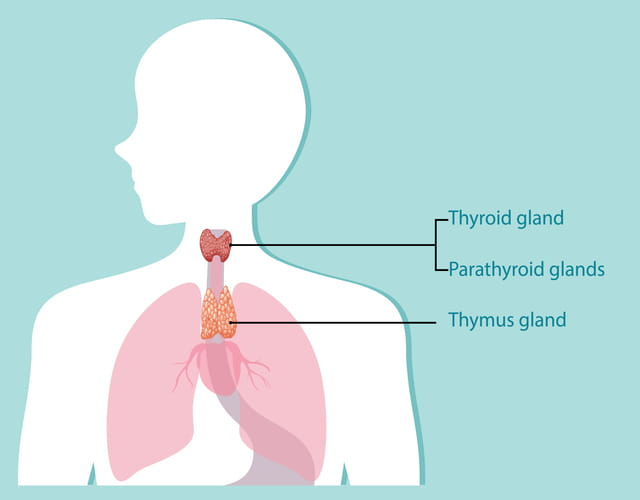
Thymic neoplasms are tumors that develop in the thymus gland. The thymus gland is an organ in the lymphatic system. Located in the chest behind the breastbone, the thymus creates T lymphocytes or white blood cells that help protect the body against infections.
People with thymic neoplasms also frequently have myasthenia gravis — a chronic, progressive autoimmune disorder that causes the muscles of the head, face, and body to become weak and easily fatigued.
On this page:
What Are Thymic Neoplasms and Myasthenia Gravis?
What are thymic neoplasms?
Thymic neoplasms are tumors that develop in the thymus gland. The thymus gland is part of the lymphatic system. It sits in the upper chest, just behind the sternum (breastbone). It makes lymphocytes (white blood cells) that protect against infection.
The thymus gland is most active from early childhood until adolescence, when it gradually shrinks and is no longer needed. But, for reasons not fully understood, the thymus is known to play a role in myasthenia gravis – a chronic, progressive autoimmune disorder.
People with thymic neoplasms often have myasthenia gravis — a disorder that causes the muscles of the head, face, and body to become weak and easily fatigued.
What are the types of thymic neoplasms?
Different types of tumors can develop in the thymus. Doctors refer to these as thymic neoplasms.
Types of thymic neoplasms include:
- Thymomas — The most common tumor of the thymus involving the cells on the outside surface of the thymus gland. Thymomas grow slowly and rarely metastasize (spread) to other body parts. Thymoma tumor cells have a similar appearance to normal thymus gland cells. Thymomas are easier to treat than thymic carcinomas.
- Thymic carcinomas — A rare thymic gland cancer with cells that look radically different from normal thymus gland cells. This type of thymic neoplasm grows more rapidly and frequently spreads to other parts of the body by the time the cancer is detected. Thymic carcinomas are more difficult to treat than thymomas.
People with thymic neoplasms frequently have an autoimmune disease, such as myasthenia gravis, that causes the body’s immune system to attack healthy tissues and organs.
What Is myasthenia gravis?
Myasthenia gravis is a rare neuromuscular autoimmune disease that causes the body’s immune system to attack healthy organs and tissues, resulting in their destruction.
MG blocks the brain impulses that travel through the nerves before they reach the muscles. The body essentially attacks itself, stopping muscle function. It can affect a person’s ability to breathe, see, smile, swallow, walk, and participate in tasks of daily living.
MG can be difficult to diagnose because each person’s MG is different. Symptoms often change from day to day, week to week, and month to month. That’s why MG is sometimes called a “snowflake disease.”
Because of varying symptoms, some people go for years without the proper diagnosis. It can occur in people of any age, but commonly occurs in women under age 40 and men over age 60.
How common is myasthenia gravis?
Myasthenia gravis is rare. It is estimated that 37 out of every 100,000 people have myasthenia gravis in the U.S. While the disorder can affect people of all ages, women under 50 are more likely to have MG compared to men. On the other hand, there are more cases of men ages 65 and up with MG than women with MG in the same age group. Although children can be diagnosed with MG, it’s much more common in adults.
In addition, although people of African descent are more prone to MG, it can affect people of all races and ethnicities.
What causes myasthenia gravis?
Normally, your body’s nerves send your muscles messages through a receptor. Nerves use a chemical called acetylcholine to deliver these messages. When the acetylcholine binds to the receptor area of a nerve, the muscle contracts.
With myasthenia gravis, the immune system attacks the muscle's receptor for acetylcholine.
Experts don’t understand exactly why the body begins to produce immune antibodies that attack acetylcholine receptors. However, it seems to relate in some way to the thymus gland, which assists the body in producing antibodies.
Approximately 15% of people with myasthenia gravis also have thymomas.
How is myasthenia gravis classified?
MG is divided into five main classes based on its clinical features and severity.
The five main classes are:
- Class I – Only eye muscles are affected; no other muscle groups are affected.
- Class II – Mild weakness of other muscles with eye muscle weakness of any severity.
- Class III – Moderate weakness of other muscles with eye muscle weakness of any severity.
- Class IV – Severe weakness of other muscles with eye muscle weakness of any severity.
- Class V – Requires intubation with or without mechanical ventilation.
Each class has a different prognosis and expected response to therapy.
Myasthenia gravis risk factors
General risk factors for myasthenia gravis include:
- Being a man between the ages of 60 to 70.
- Being a woman between the ages of 20 to 30.
- Being born to a mother whose abnormal antibodies entered your body through the placenta during pregnancy.
- Having specific genetic markers called HLA-B8 or DR3.
Lifestyle risk factors for MG include:
- Being obese.
- Using illicit drugs.
- Smoking.
- Lack of physical activity.
- Poor diet (including low consumption of fish).
In 5% of those with myasthenia gravis, there is a family member with some type of autoimmune disorder.
Complications of myasthenia gravis
Left untreated, myasthenia gravis can lead to serious complications, including:
- Difficulty walking.
- Difficulty breathing.
- Respiratory distress.
- Slurred speech.
- Widespread pain.
It’s important to manage MG with appropriate medical treatment to prevent these complications and maintain your quality of life.
How can I prevent MG?
MG can’t be prevented because its causes are unknown. However, if you’ve been diagnosed with myasthenia gravis, you may be able to control your flare-ups through lifestyle changes.
To prevent MG flare-ups:
- Avoid strenuous activities.
- Get adequate sleep.
- Manage your stress.
- Rest when needed.
- Stay away from sick people.
- Stay out of extreme heat and cold.
- Work with your provider to change medications that cause flare-ups.
What Are the Signs and Symptoms of Thymic Neoplasms and Myasthenia Gravis?
Symptoms of thymic neoplasms
Frequently, thymic neoplasms do not cause any recognizable symptoms. Doctors may discover the cancer during routine chest x-rays. But several conditions could indicate the presence of thymic neoplasms.
If you experience any of the following symptoms, consult your doctor:
- Chest pain.
- Difficulty breathing.
- Persistent, chronic cough.
Symptoms of myasthenia gravis
The primary sign that a person may have myasthenia gravis is muscle weakness that lessens when you rest.
Common symptoms include:
- Altered speech — soft or nasal-sounding speech.
- Choking easily while eating.
- Difficulty chewing because of muscle fatigue.
- Difficulty swallowing.
- Double vision.
- Drooping eyelids — one or both eyes.
- Fewer than normal facial expressions
- Weakness in the neck or arms.
When should I see a doctor about my myasthenia gravis symptoms?
If you have any of the symptoms of MG above, call your doctor promptly. Early and proper diagnosis and treatment can prevent further muscle weakening.
How Do You Diagnose Thymic Neoplasms and Myasthenia Gravis?
Diagnosing thymic neoplasms
Your doctor will conduct a thorough physical exam to check for signs of disease or anything that appears unusual. They will likely take a history of your health background, lifestyle habits, past illnesses, and treatments.
Your health care provider may use one of the following tests or procedures to detect and identify a thymoma or thymic carcinoma:
- Chest x-ray — Uses radiation to take pictures of the bones and organs inside your chest.
- CT scan— Uses a computer to take a series of detailed pictures of certain areas inside your body from several different angles. You may need to have dye injected into a vein or take it orally to make the organs and tissues inside show up more clearly.
- Magnetic resonance imaging (MRI) — uses a powerful magnet, radio waves, and a computer to take a series of pictures of areas inside the body.
- Positron emission tomography scan (PET scan) — requires that you have a small amount of radioactive glucose injected into your vein. The PET scanning device rotates around your body, taking pictures. Cancer cells show up as bright spots in the picture because they take up more glucose than normal cells do.
Diagnosing myasthenia gravis
Your doctor may conduct one or more tests to confirm a diagnosis of myasthenia gravis:
- Blood panel — to check for certain abnormal antibodies.
- Edrophonium test — injection of the chemical edrophonium. If this results in a sudden, yet temporary, improvement in muscle strength and stamina, it’s a good indicator that you may have myasthenia gravis.
- Imaging scans — CT scan or MRI to check for abnormal size or appearance of the thymus gland.
- Repetitive nerve stimulation — to test how muscles respond to nerve stimulation.
- Single-fiber electromyography (EMG) — to check the health of the muscles and the nerves that control the muscles.
How Do You Treat Myasthenia Gravis?
Nonsurgical myasthenia gravis treatments
For mild cases of myasthenia gravis — or for people over the age of 60 — doctors can use the following nonsurgical treatments to relieve symptoms:
- Acetylcholinesterase inhibitors – These drugs prevent the breakdown of acetylcholine, which boosts the amount of acetylcholine available to bind to the receptors. This, in turn, improves muscle activation by increasing the signals between muscles and nerves. Acetylcholinesterase inhibitors are a first-line treatment for mild MG symptoms.
- Corticosteroids and other nonsteroidal immunosuppressants – These drugs reduce the body’s production of certain white blood cells, which also limits the production of abnormal antibodies. The fewer abnormal antibodies produced, the better the body’s acetylcholine receptors work, which aids communication between nerves and muscles and improves muscle activation. Steroids and immunosuppressants are effective for 70% of users, who report significant improvement in MG symptoms. They are usually the next lines of treatment if acetylcholinesterase inhibitors do not work.
- Plasmapheresis – Also known as therapeutic plasma exchange, plasmapheresis separates blood into its different components to remove the blood plasma containing the abnormal antibodies causing MG symptoms. The blood is then returned to the body without the antibodies, which should improve communication between the nerves and muscles. This procedure can be used as a short-term treatment in an emergency or when other treatments are not working and a quick boost in strength is needed, such as before surgery. It can be repeated as needed.
- Intravenous immunoglobulin – Immunoglobulins help the body fight infection and disease. This treatment increases the amount of immunoglobulin in your blood, which reduces the levels of abnormal antibodies causing MG symptoms. Also used in emergencies or when a quick improvement in strength is needed, it can be given as an IV drip over two to five days in an emergency or every three to six weeks as a maintenance drug.
- Targeted treatments — Recent advances have been made in the treatment of myasthenia gravis with medications that target specific antibodies and proteins in the body. B-cell-directed drugs, neonatal Fc receptor blockers, and complement inhibitors are examples of targeted therapies aimed at reducing the symptoms of MG.
Surgical myasthenia gravis treatments
Thymectomy surgery
If you have a thymic tumor, with or without myasthenia gravis, you will need to undergo thymectomy surgery to remove your thymus gland. Even if you don’t have a thymic neoplasm, your doctor may advise you to undergo a thymectomy for myasthenia gravis.
Many health care facilities offer only open chest surgery for thymectomy procedures. Thoracic surgeons at the UPMC perform both minimally invasive and robotic-assisted thymectomies, in addition to traditional open chest surgery.
During a thymectomy, the surgeon will remove the affected part of the thymus or the entire thymus gland.
Even if the entire thymic tumor is removed or a thymectomy is performed, the surgeon may order radiation therapy after surgery to make sure no more cancer cells develop. The radiation will lower your risk of the cancer returning.
Other adjuvant therapies for thymic neoplasms may include chemotherapy and hormone therapy.
















